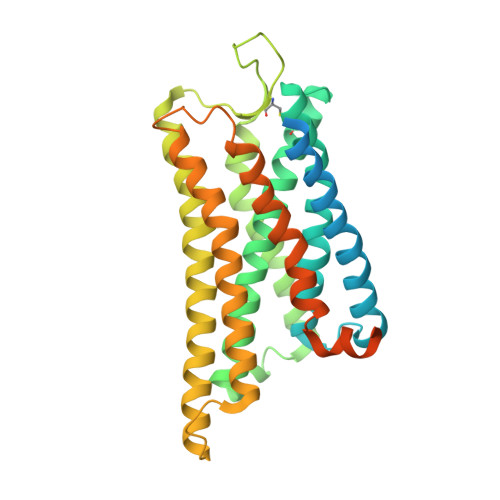
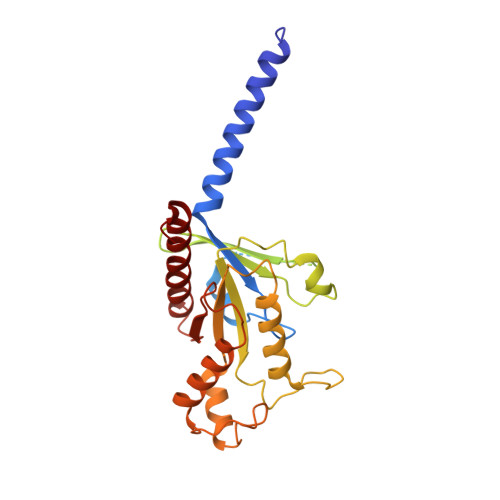
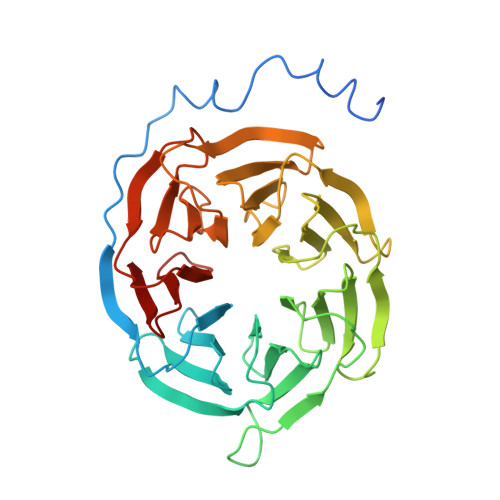
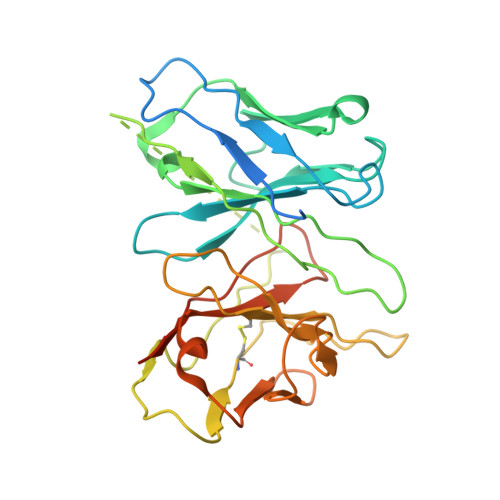
Structural insights into ligand recognition and selectivity of somatostatin receptors.
Zhao, W., Han, S., Qiu, N., Feng, W., Lu, M., Zhang, W., Wang, M., Zhou, Q., Chen, S., Xu, W., Du, J., Chu, X., Yi, C., Dai, A., Hu, L., Shen, M.Y., Sun, Y., Zhang, Q., Ma, Y., Zhong, W., Yang, D., Wang, M.W., Wu, B., Zhao, Q.(2022) Cell Res 32: 761-772
- PubMed: 35739238
- DOI: https://doi.org/10.1038/s41422-022-00679-x
- Primary Citation of Related Structures:
7XMR, 7XMS, 7XMT, 7XN9, 7XNA - PubMed Abstract:
Somatostatin receptors (SSTRs) play versatile roles in inhibiting the secretion of multiple hormones such as growth hormone and thyroid-stimulating hormone, and thus are considered as targets for treating multiple tumors. Despite great progress made in therapeutic development against this diverse receptor family, drugs that target SSTRs still show limited efficacy with preferential binding affinity and conspicuous side-effects. Here, we report five structures of SSTR2 and SSTR4 in different states, including two crystal structures of SSTR2 in complex with a selective peptide antagonist and a non-peptide agonist, respectively, a cryo-electron microscopy (cryo-EM) structure of G i1 -bound SSTR2 in the presence of the endogenous ligand SST-14, as well as two cryo-EM structures of G i1 -bound SSTR4 in complex with SST-14 and a small-molecule agonist J-2156, respectively. By comparison of the SSTR structures in different states, molecular mechanisms of agonism and antagonism were illustrated. Together with computational and functional analyses, the key determinants responsible for ligand recognition and selectivity of different SSTR subtypes and multiform binding modes of peptide and non-peptide ligands were identified. Insights gained in this study will help uncover ligand selectivity of various SSTRs and accelerate the development of new molecules with better efficacy by targeting SSTRs.
Organizational Affiliation:
State Key Laboratory of Drug Research and CAS Key Laboratory of Receptor Research, Shanghai Institute of Materia Medica, Chinese Academy of Sciences, Shanghai, China.