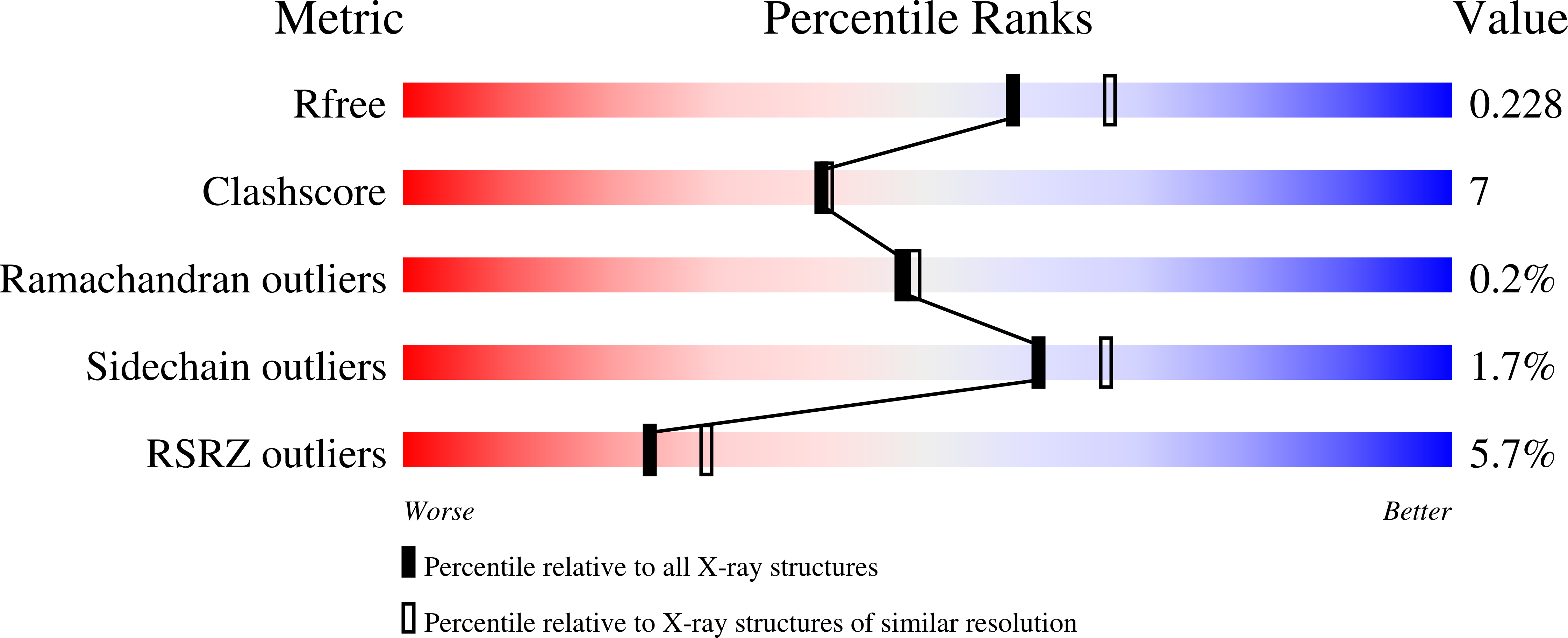
Disruption of the coenzyme binding site and dimer interface revealed in the crystal structure of mitochondrial aldehyde dehydrogenase "asian" variant
Larson, H.N., Weiner, H., Hurley, T.D.(2005) J Biol Chem 280: 30550-30556
- PubMed: 15983043
- DOI: https://doi.org/10.1074/jbc.M502345200
- Primary Citation of Related Structures:
1ZUM - PubMed Abstract:
Mitochondrial aldehyde dehydrogenase (ALDH2) is the major enzyme that oxidizes ethanol-derived acetaldehyde. A nearly inactive form of the enzyme, ALDH2*2, is found in about 40% of the East Asian population. This variant enzyme is defined by a glutamate to lysine substitution at residue 487 located within the oligomerization domain. ALDH2*2 has an increased Km for its coenzyme, NAD+, and a decreased kcat, which lead to low activity in vivo. Here we report the 2.1 A crystal structure of ALDH2*2. The structure shows a large disordered region located at the dimer interface that includes much of the coenzyme binding cleft and a loop of residues that form the base of the active site. As a consequence of these structural changes, the variant enzyme exhibits rigid body rotations of its catalytic and coenzyme-binding domains relative to the oligomerization domain. These structural perturbations are the direct result of the inability of lysine 487 to form important stabilizing hydrogen bonds with arginines 264 and 475. Thus, the elevated Km for coenzyme exhibited by this variant probably reflects the energetic penalty for reestablishing this site for productive coenzyme binding, whereas the structural alterations near the active site are consistent with the lowered Vmax.
Organizational Affiliation:
Department of Biochemistry and Molecular Biology, Indiana University School of Medicine, Indianapolis, Indiana 46202, USA.