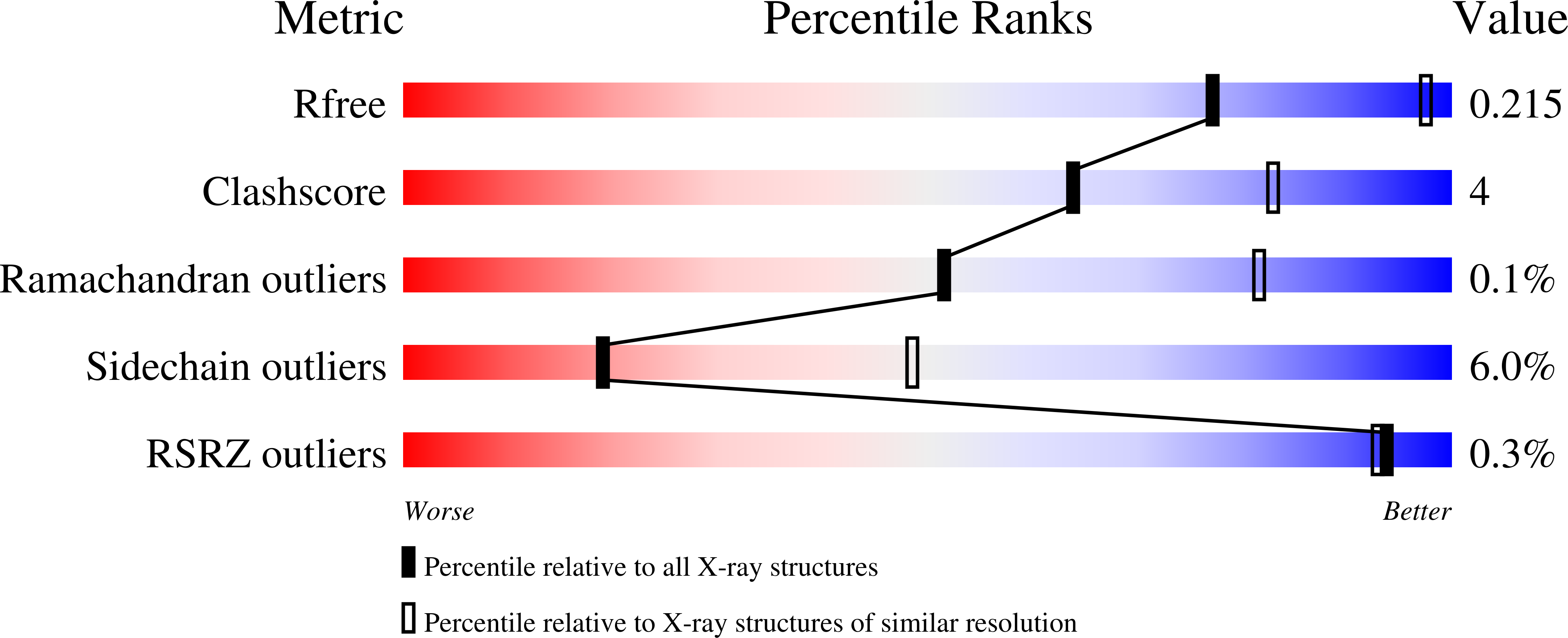
Joint functions of protein residues and NADP(H) in oxygen activation by flavin-containing monooxygenase.
Orru, R., Pazmino, D.E., Fraaije, M.W., Mattevi, A.(2010) J Biol Chem 285: 35021-35028
- PubMed: 20807767
- DOI: https://doi.org/10.1074/jbc.M110.161372
- Primary Citation of Related Structures:
2XLP, 2XLR, 2XLS, 2XLT, 2XLU - PubMed Abstract:
The reactivity of flavoenzymes with dioxygen is at the heart of a number of biochemical reactions with far reaching implications for cell physiology and pathology. Flavin-containing monooxygenases are an attractive model system to study flavin-mediated oxygenation. In these enzymes, the NADP(H) cofactor is essential for stabilizing the flavin intermediate, which activates dioxygen and makes it ready to react with the substrate undergoing oxygenation. Our studies combine site-directed mutagenesis with the usage of NADP(+) analogues to dissect the specific roles of the cofactors and surrounding protein matrix. The highlight of this "double-engineering" approach is that subtle alterations in the hydrogen bonding and stereochemical environment can drastically alter the efficiency and outcome of the reaction with oxygen. This is illustrated by the seemingly marginal replacement of an Asn to Ser in the oxygen-reacting site, which inactivates the enzyme by effectively converting it into an oxidase. These data rationalize the effect of mutations that cause enzyme deficiency in patients affected by the fish odor syndrome. A crucial role of NADP(+) in the oxygenation reaction is to shield the reacting flavin N5 atom by H-bond interactions. A Tyr residue functions as backdoor that stabilizes this crucial binding conformation of the nicotinamide cofactor. A general concept emerging from this analysis is that the two alternative pathways of flavoprotein-oxygen reactivity (oxidation versus monooxygenation) are predicted to have very similar activation barriers. The necessity of fine tuning the hydrogen-bonding, electrostatics, and accessibility of the flavin will represent a challenge for the design and development of oxidases and monoxygenases for biotechnological applications.
Organizational Affiliation:
Department of Genetics and Microbiology, University of Pavia, Via Ferrata 1, 27100 Pavia, Italy.