The crystal structure of a glycosyl hydrolases (GH) family protein 16 from Mycobacterium smegmatis str. MC2 155
Tan, K., Chhor, G., Bearden, J., Joachimiak, A.To be published.
Experimental Data Snapshot
wwPDB Validation 3D Report Full Report
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Glycosyl hydrolases family protein 16 | 269 | Mycolicibacterium smegmatis MC2 155 | Mutation(s): 0 Gene Names: MSMEG_5345 | 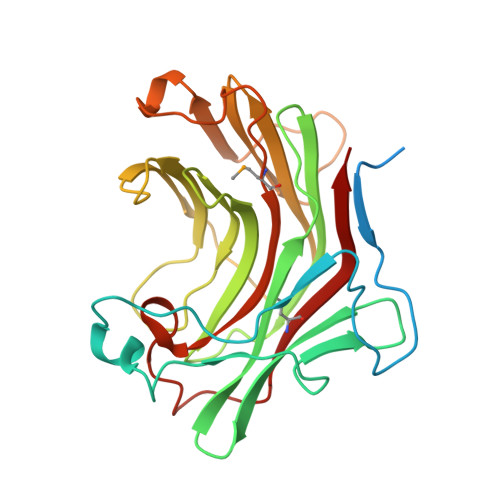 | |
UniProt | |||||
Find proteins for A0R351 (Mycolicibacterium smegmatis (strain ATCC 700084 / mc(2)155)) Explore A0R351 Go to UniProtKB: A0R351 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A0R351 | ||||
Sequence AnnotationsExpand | |||||
| |||||
| Ligands 6 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| PG4 Query on PG4 | F [auth A], G [auth A], H [auth A], I [auth A], J [auth A] | TETRAETHYLENE GLYCOL C8 H18 O5 UWHCKJMYHZGTIT-UHFFFAOYSA-N |  | ||
| 211 Query on 211 | P [auth A] | 2,2',2''-NITRILOTRIETHANOL C6 H15 N O3 GSEJCLTVZPLZKY-UHFFFAOYSA-N |  | ||
| PEG Query on PEG | D [auth A], E [auth A] | DI(HYDROXYETHYL)ETHER C4 H10 O3 MTHSVFCYNBDYFN-UHFFFAOYSA-N |  | ||
| PO4 Query on PO4 | C [auth A] | PHOSPHATE ION O4 P NBIIXXVUZAFLBC-UHFFFAOYSA-K |  | ||
| GOL Query on GOL | K [auth A], L [auth A], M [auth A], N [auth A], O [auth A] | GLYCEROL C3 H8 O3 PEDCQBHIVMGVHV-UHFFFAOYSA-N |  | ||
| CA Query on CA | B [auth A] | CALCIUM ION Ca BHPQYMZQTOCNFJ-UHFFFAOYSA-N |  | ||
| Modified Residues 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Type | Formula | 2D Diagram | Parent |
| MSE Query on MSE | A | L-PEPTIDE LINKING | C5 H11 N O2 Se |  | MET |
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 117.42 | α = 90 |
| b = 117.42 | β = 90 |
| c = 54.909 | γ = 90 |
| Software Name | Purpose |
|---|---|
| SBC-Collect | data collection |
| SHELXD | phasing |
| MLPHARE | phasing |
| DM | model building |
| ARP | model building |
| WARP | model building |
| HKL-3000 | phasing |
| PHENIX | refinement |
| HKL-3000 | data reduction |
| HKL-3000 | data scaling |
| DM | phasing |