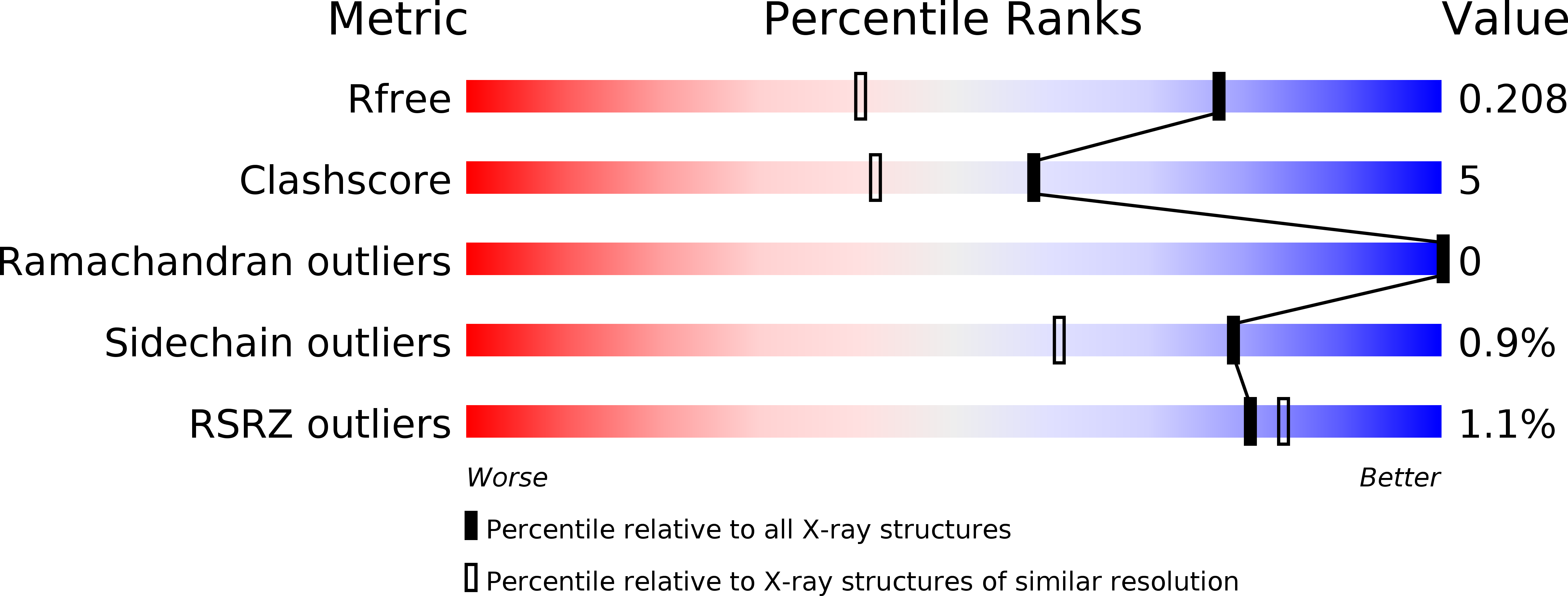
Structural Insights into the Development of Cycloguanil Derivatives asTrypanosoma bruceiPteridine-Reductase-1 Inhibitors.
Landi, G., Linciano, P., Borsari, C., Bertolacini, C.P., Moraes, C.B., Cordeiro-da-Silva, A., Gul, S., Witt, G., Kuzikov, M., Costi, M.P., Pozzi, C., Mangani, S.(2019) ACS Infect Dis 5: 1105-1114
- PubMed: 31012301
- DOI: https://doi.org/10.1021/acsinfecdis.8b00358
- Primary Citation of Related Structures:
6HNC, 6HNR, 6HOW - PubMed Abstract:
Cycloguanil is a known dihydrofolate-reductase (DHFR) inhibitor, but there is no evidence of its activity on pteridine reductase (PTR), the main metabolic bypass to DHFR inhibition in trypanosomatid parasites. Here, we provide experimental evidence of cycloguanil as an inhibitor of Trypanosoma brucei PTR1 ( Tb PTR1). A small library of cycloguanil derivatives was developed, resulting in 1 and 2a having IC 50 values of 692 and 186 nM, respectively, toward Tb PTR1. Structural analysis revealed that the increased potency of 1 and 2a is due to the combined contributions of hydrophobic interactions, H-bonds, and halogen bonds. Moreover, in vitro cell-growth-inhibition tests indicated that 2a is also effective on T. brucei . The simultaneous inhibition of DHFR and PTR1 activity in T. brucei is a promising new strategy for the treatment of human African trypanosomiasis. For this purpose, 1,6-dihydrotriazines represent new molecular tools to develop potent dual PTR and DHFR inhibitors.
Organizational Affiliation:
Department of Biotechnology, Chemistry and Pharmacy-Department of Excellence 2018-2020 , University of Siena , via Aldo Moro 2 , 53100 Siena , Italy.