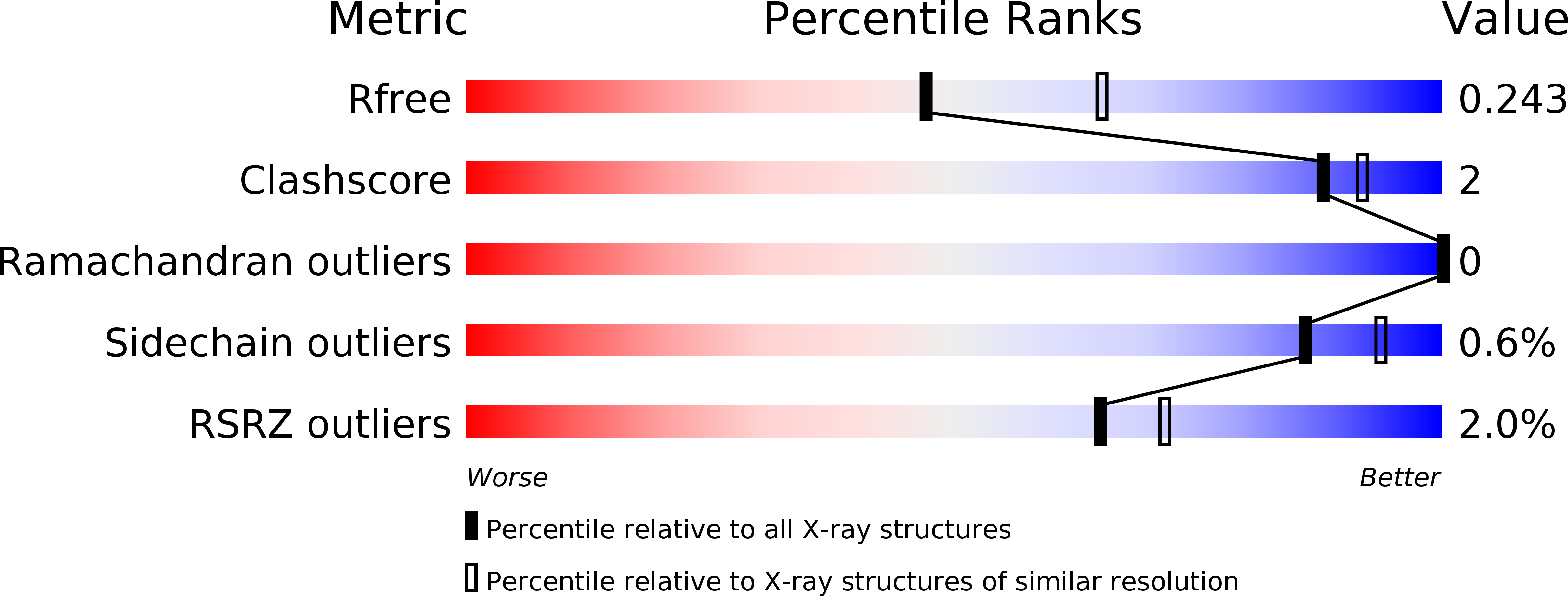
Structural Basis and Evolution of Glycan Receptor Specificities within the Polyomavirus Family.
Stroh, L.J., Rustmeier, N.H., Blaum, B.S., Botsch, J., Rossler, P., Wedekink, F., Lipkin, W.I., Mishra, N., Stehle, T.(2020) mBio 11
- PubMed: 32723915
- DOI: https://doi.org/10.1128/mBio.00745-20
- Primary Citation of Related Structures:
6Y5X, 6Y5Y, 6Y5Z, 6Y60, 6Y61, 6Y63, 6Y64, 6Y65, 6Y66, 6Y67, 6Y6A, 6Y9I - PubMed Abstract:
Asymptomatic infections with polyomaviruses in humans are common, but these small viruses can cause severe diseases in immunocompromised hosts. New Jersey polyomavirus (NJPyV) was identified via a muscle biopsy in an organ transplant recipient with systemic vasculitis, myositis, and retinal blindness, and human polyomavirus 12 (HPyV12) was detected in human liver tissue. The evolutionary origins and potential diseases are not well understood for either virus. In order to define their receptor engagement strategies, we first used nuclear magnetic resonance (NMR) spectroscopy to establish that the major capsid proteins (VP1) of both viruses bind to sialic acid in solution. We then solved crystal structures of NJPyV and HPyV12 VP1 alone and in complex with sialylated glycans. NJPyV employs a novel binding site for a α2,3-linked sialic acid, whereas HPyV12 engages terminal α2,3- or α2,6-linked sialic acids in an exposed site similar to that found in Trichodysplasia spinulosa -associated polyomavirus (TSPyV). Gangliosides or glycoproteins, featuring in mammals usually terminal sialic acids, are therefore receptor candidates for both viruses. Structural analyses show that the sialic acid-binding site of NJPyV is conserved in chimpanzee polyomavirus (ChPyV) and that the sialic acid-binding site of HPyV12 is widely used across the entire polyomavirus family, including mammalian and avian polyomaviruses. A comparison with other polyomavirus-receptor complex structures shows that their capsids have evolved to generate several physically distinct virus-specific receptor-binding sites that can all specifically engage sialylated glycans through a limited number of contacts. Small changes in each site may have enabled host-switching events during the evolution of polyomaviruses. IMPORTANCE Virus attachment to cell surface receptors is critical for productive infection. In this study, we have used a structure-based approach to investigate the cell surface recognition event for New Jersey polyomavirus (NJPyV) and human polyomavirus 12 (HPyV12). These viruses belong to the polyomavirus family, whose members target different tissues and hosts, including mammals, birds, fish, and invertebrates. Polyomaviruses are nonenveloped viruses, and the receptor-binding site is located in their capsid protein VP1. The NJPyV capsid features a novel sialic acid-binding site that is shifted in comparison to other structurally characterized polyomaviruses but shared with a closely related simian virus. In contrast, HPyV12 VP1 engages terminal sialic acids in a manner similar to the human Trichodysplasia spinulosa -associated polyomavirus. Our structure-based phylogenetic analysis highlights that even distantly related avian polyomaviruses possess the same exposed sialic acid-binding site. These findings complement phylogenetic models of host-virus codivergence and may also reflect past host-switching events.
Organizational Affiliation:
Interfaculty Institute of Biochemistry, University of Tübingen, Tübingen, Germany.