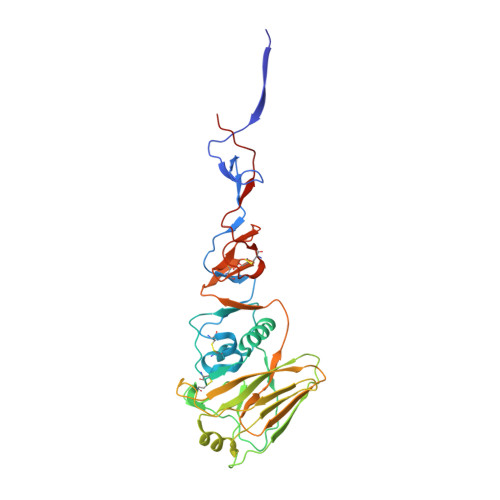
Evolution of human H3N2 influenza virus receptor specificity has substantially expanded the receptor-binding domain site.
Thompson, A.J., Wu, N.C., Canales, A., Kikuchi, C., Zhu, X., de Toro, B.F., Canada, F.J., Worth, C., Wang, S., McBride, R., Peng, W., Nycholat, C.M., Jimenez-Barbero, J., Wilson, I.A., Paulson, J.C.(2024) Cell Host Microbe 32: 261
- PubMed: 38307019
- DOI: https://doi.org/10.1016/j.chom.2024.01.003
- Primary Citation of Related Structures:
8TJ4, 8TJ6, 8TJ7, 8TJ8, 8TJ9, 8TJA, 8TJB - PubMed Abstract:
Hemagglutinins (HAs) from human influenza viruses descend from avian progenitors that bind α2-3-linked sialosides and must adapt to glycans with α2-6-linked sialic acids on human airway cells to transmit within the human population. Since their introduction during the 1968 pandemic, H3N2 viruses have evolved over the past five decades to preferentially recognize human α2-6-sialoside receptors that are elongated through addition of poly-LacNAc. We show that more recent H3N2 viruses now make increasingly complex interactions with elongated receptors while continuously selecting for strains maintaining this phenotype. This change in receptor engagement is accompanied by an extension of the traditional receptor-binding site to include residues in key antigenic sites on the surface of HA trimers. These results help explain the propensity for selection of antigenic variants, leading to vaccine mismatching, when H3N2 viruses are propagated in chicken eggs or cells that do not contain such receptors.
Organizational Affiliation:
Department of Molecular Medicine, The Scripps Research Institute, La Jolla, CA 92037, USA.