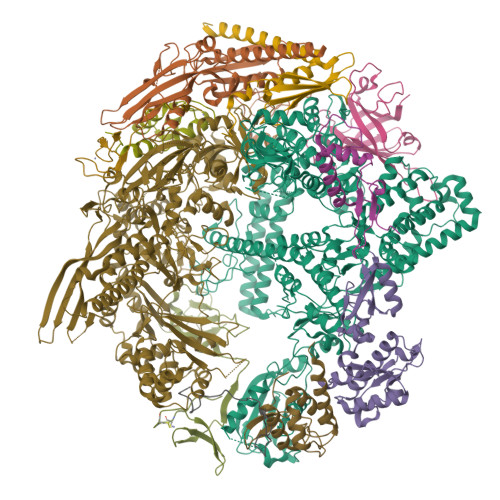
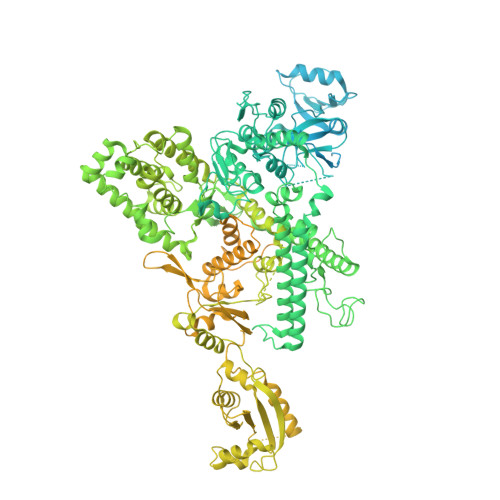
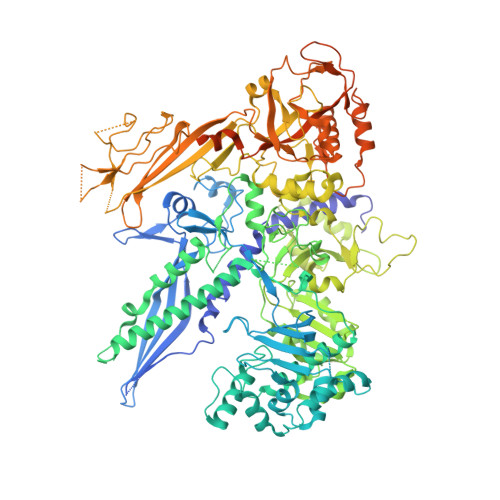
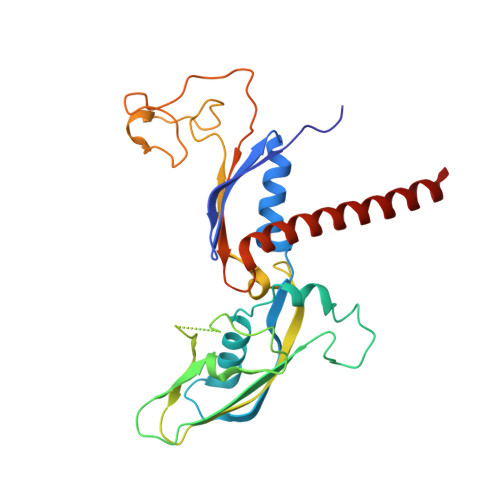
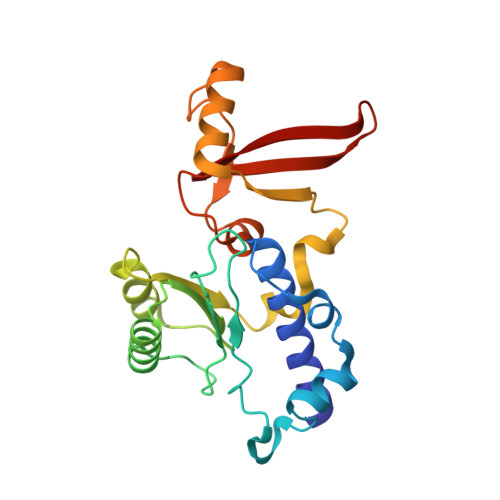
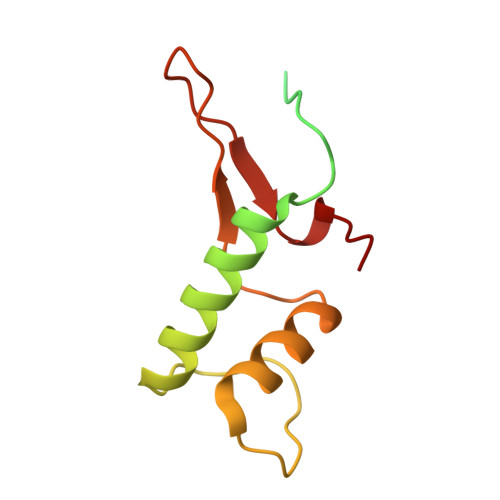
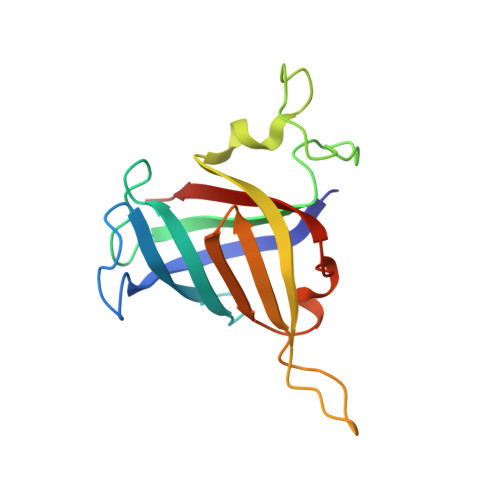
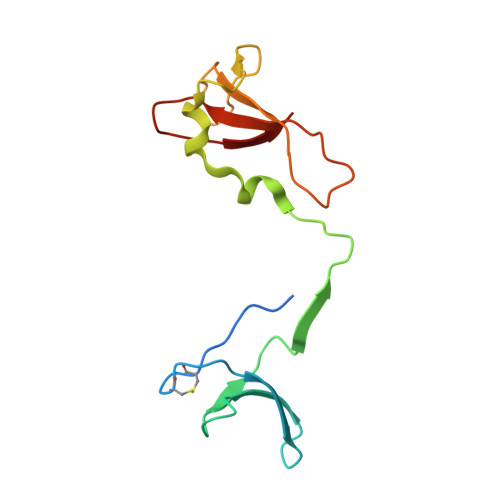
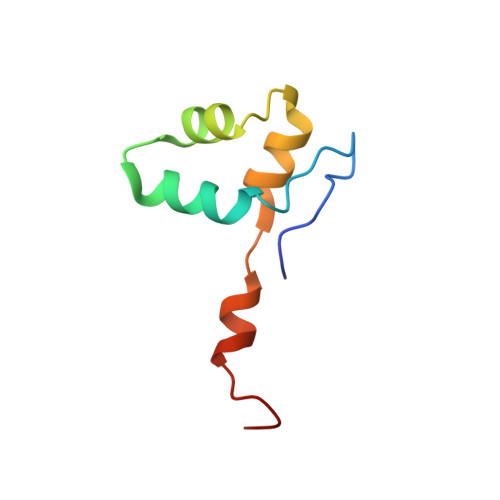
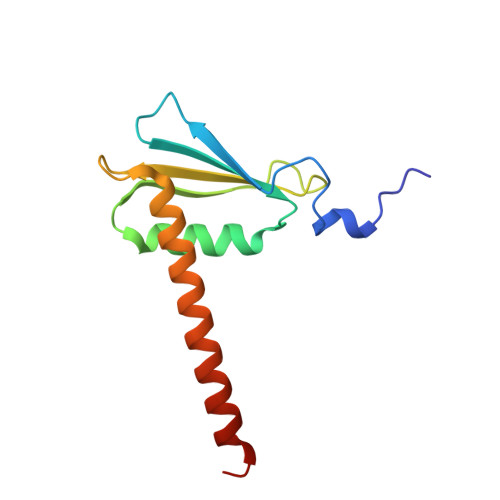
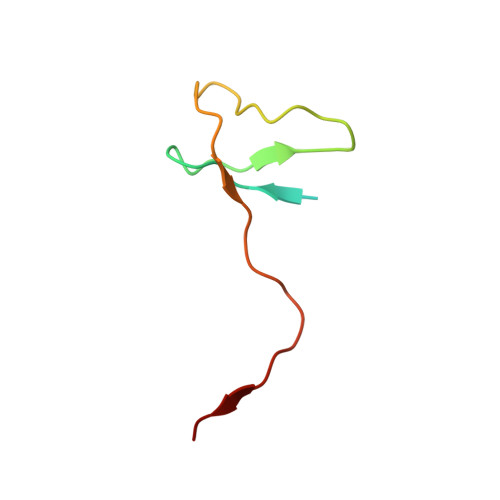
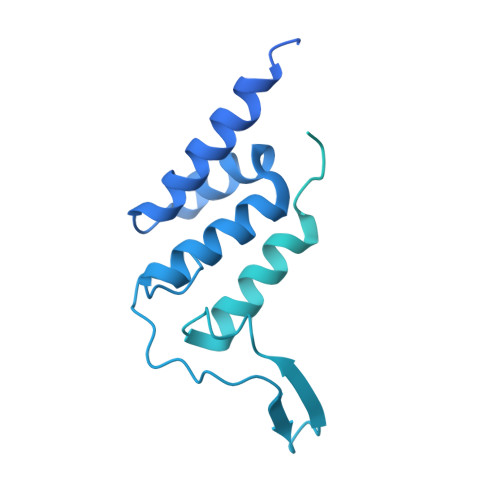
Cryo-EM structure of mammalian RNA polymerase II in complex with human RPAP2.
Fianu, I., Dienemann, C., Aibara, S., Schilbach, S., Cramer, P.(2021) Commun Biol 4: 606-606
- PubMed: 34021257
- DOI: https://doi.org/10.1038/s42003-021-02088-z
- Primary Citation of Related Structures:
7B7U - PubMed Abstract:
Nuclear import of RNA polymerase II (Pol II) involves the conserved factor RPAP2. Here we report the cryo-electron microscopy (cryo-EM) structure of mammalian Pol II in complex with human RPAP2 at 2.8 Å resolution. The structure shows that RPAP2 binds between the jaw domains of the polymerase subunits RPB1 and RPB5. RPAP2 is incompatible with binding of downstream DNA during transcription and is displaced upon formation of a transcription pre-initiation complex.
Organizational Affiliation:
Department of Molecular Biology, Max Planck Institute for Biophysical Chemistry, Göttingen, Germany.