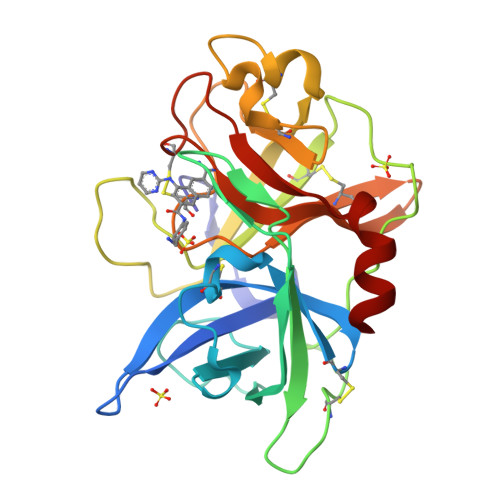
Interaction with the S1beta-pocket of urokinase: 8-heterocycle substituted and 6,8-disubstituted 2-naphthamidine urokinase inhibitors.
Wendt, M.D., Geyer, A., McClellan, W.J., Rockway, T.W., Weitzberg, M., Zhao, X., Mantei, R., Stewart, K., Nienaber, V., Klinghofer, V., Giranda, V.L.(2004) Bioorg Med Chem Lett 14: 3063-3068
- PubMed: 15149645
- DOI: https://doi.org/10.1016/j.bmcl.2004.04.030
- Primary Citation of Related Structures:
1SQA, 1SQO, 1SQT - PubMed Abstract:
Several 8-substituted 2-naphthamidine-based inhibitors of the serine protease urokinase plasminogen activator (uPA) are described. Direct attachment of five-membered saturated or unsaturated rings improved inhibitor performance; substitution with sulfones further improved binding profiles. Combination of these substituents or of previously described NH-linked heteroaromatic rings with 6-phenyl amide substituents provided further enhancements to potency and selectivity.
Organizational Affiliation:
Cancer Research, Global Pharmaceutical R & D, Abbott Laboratories, 100 Abbott Park Road, Abbott Park, IL 60064-6101, USA. mike.d.wendt@abbott.com