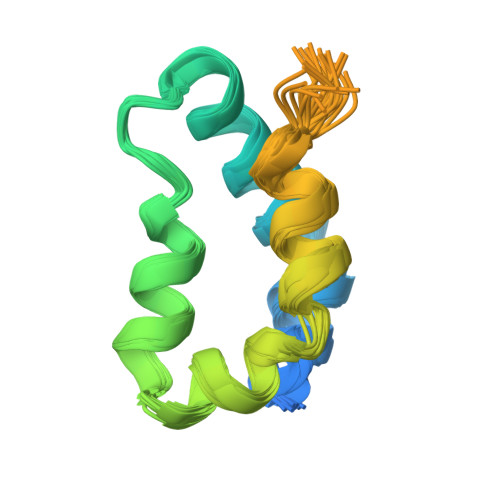
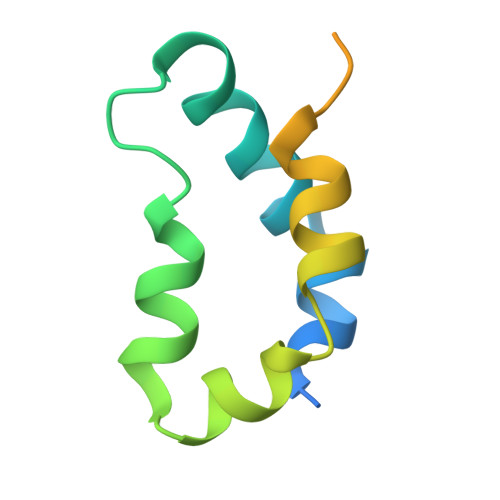
Structure of the XPC binding domain of hHR23A reveals hydrophobic patches for protein interaction
Kamionka, M., Feigon, J.(2004) Protein Sci 13: 2370-2377
- PubMed: 15322280
- DOI: https://doi.org/10.1110/ps.04824304
- Primary Citation of Related Structures:
1TP4 - PubMed Abstract:
Rad23 proteins are involved both in the ubiquitin-proteasome pathway and in nucleotide excision repair (NER), but the relationship between these two pathways is not yet understood. The two human homologs of Rad23, hHR23A and B, are functionally redundant in NER and interact with xeroderma pigmentosum complementation group C (XPC) protein. The XPC-hHR23 complex is responsible for the specific recognition of damaged DNA, which is an early step in NER. The interaction of the XPC binding domain (XPCB) of hHR23A/B with XPC protein has been shown to be important for its optimal function in NER. We have determined the solution structure of XPCB of hHR23A. The domain consists of five amphipathic helices and reveals hydrophobic patches on the otherwise highly hydrophilic domain surface. The patches are predicted to be involved in interaction with XPC. The XPCB domain has limited sequence homology with any proteins outside of the Rad23 family except for sacsin, a protein involved in spastic ataxia of Charlevoix-Saguenay, which contains a domain with 35% sequence identity.
Organizational Affiliation:
Department of Chemistry and Biochemistry, 405 Hilgard Avenue, P.O. Box 951569, University of California, Los Angeles, California 90095-1569, USA.