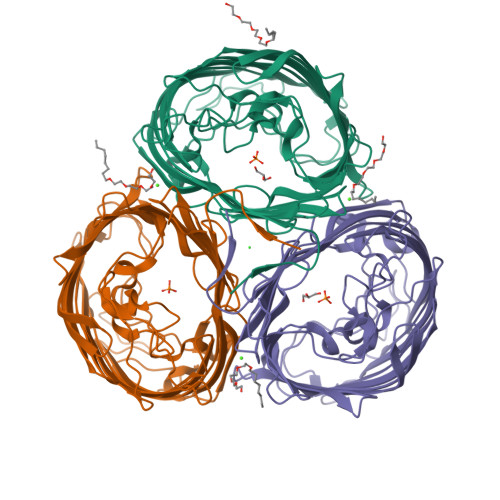
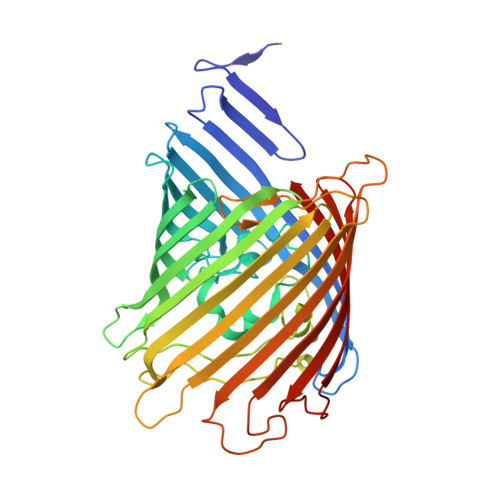
An arginine ladder in OprP mediates phosphate-specific transfer across the outer membrane.
Moraes, T.F., Bains, M., Hancock, R.E., Strynadka, N.C.(2007) Nat Struct Mol Biol 14: 85-87
- PubMed: 17187075
- DOI: https://doi.org/10.1038/nsmb1189
- Primary Citation of Related Structures:
2O4V - PubMed Abstract:
The outer membrane protein OprP mediates the transport of essential phosphate anions into the pathogenic bacterium Pseudomonas aeruginosa. Here we report the crystallographic structure of trimeric OprP at 1.9-A resolution, revealing an unprecedented 9-residue arginine 'ladder' that spans from the extracellular surface down through a constriction zone where phosphate is coordinated. Lysine residues coat the inner periplasmic surface, creating an 'electropositive sink' that pulls the phosphates through the eyelet and into the cell.
Organizational Affiliation:
Department of Biochemistry and Molecular Biology and the Center for Blood Research, University of British Columbia, 2350 Health Sciences Mall, Vancouver, British Columbia V6T 1Z3, Canada.