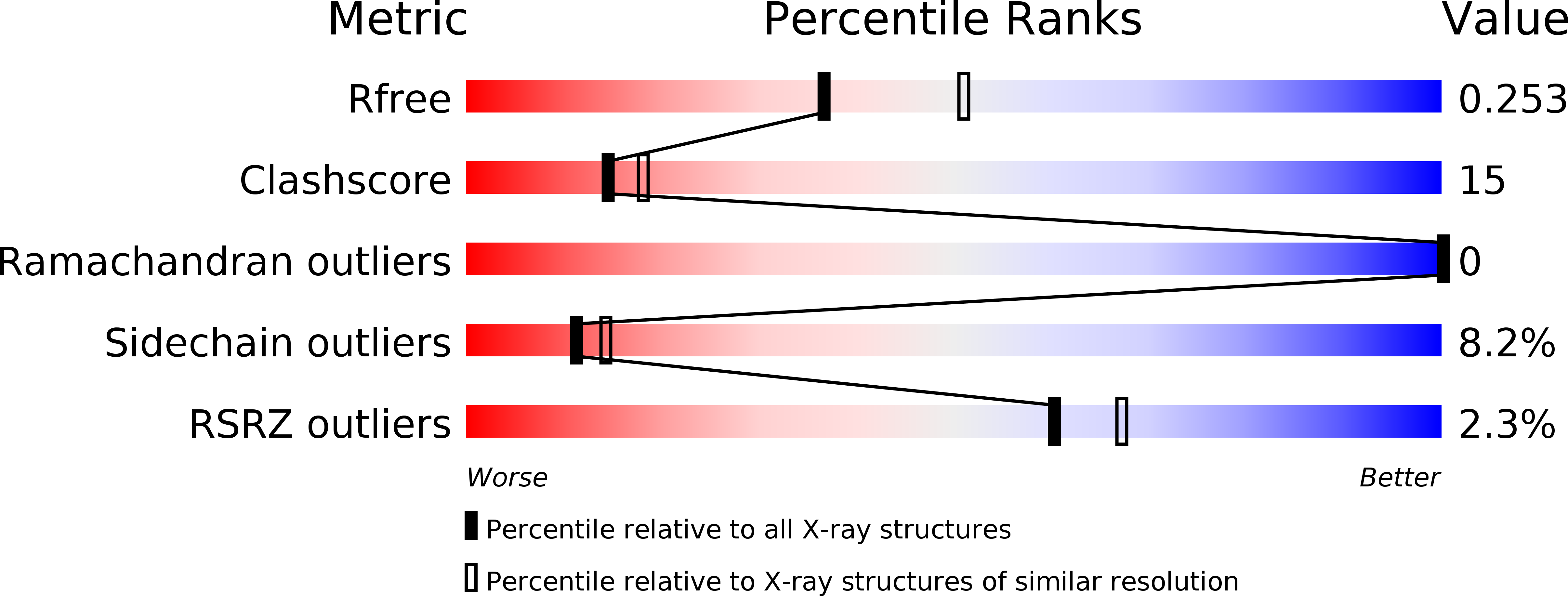
Functional and structural characterization of four glutaminases from Escherichia coli and Bacillus subtilis.
Brown, G., Singer, A., Proudfoot, M., Skarina, T., Kim, Y., Chang, C., Dementieva, I., Kuznetsova, E., Gonzalez, C.F., Joachimiak, A., Savchenko, A., Yakunin, A.F.(2008) Biochemistry 47: 5724-5735
- PubMed: 18459799
- DOI: https://doi.org/10.1021/bi800097h
- Primary Citation of Related Structures:
1MKI, 1U60, 3BRM - PubMed Abstract:
Glutaminases belong to the large superfamily of serine-dependent beta-lactamases and penicillin-binding proteins, and they catalyze the hydrolytic deamidation of L-glutamine to L-glutamate. In this work, we purified and biochemically characterized four predicted glutaminases from Escherichia coli (YbaS and YneH) and Bacillus subtilis (YlaM and YbgJ). The proteins demonstrated strict specificity to L-glutamine and did not hydrolyze D-glutamine or L-asparagine. In each organism, one glutaminase showed higher affinity to glutamine ( E. coli YbaS and B. subtilis YlaM; K m 7.3 and 7.6 mM, respectively) than the second glutaminase ( E. coli YneH and B. subtilis YbgJ; K m 27.6 and 30.6 mM, respectively). The crystal structures of the E. coli YbaS and the B. subtilis YbgJ revealed the presence of a classical beta-lactamase-like fold and conservation of several key catalytic residues of beta-lactamases (Ser74, Lys77, Asn126, Lys268, and Ser269 in YbgJ). Alanine replacement mutagenesis demonstrated that most of the conserved residues located in the putative glutaminase catalytic site are essential for activity. The crystal structure of the YbgJ complex with the glutaminase inhibitor 6-diazo-5-oxo- l-norleucine revealed the presence of a covalent bond between the inhibitor and the hydroxyl oxygen of Ser74, providing evidence that Ser74 is the primary catalytic nucleophile and that the glutaminase reaction proceeds through formation of an enzyme-glutamyl intermediate. Growth experiments with the E. coli glutaminase deletion strains revealed that YneH is involved in the assimilation of l-glutamine as a sole source of carbon and nitrogen and suggested that both glutaminases (YbaS and YneH) also contribute to acid resistance in E. coli.
Organizational Affiliation:
Banting and Best Department of Medical Research, Ontario Centre for Structural Proteomics, University of Toronto, Toronto, Ontario M5G 1L6, Canada.