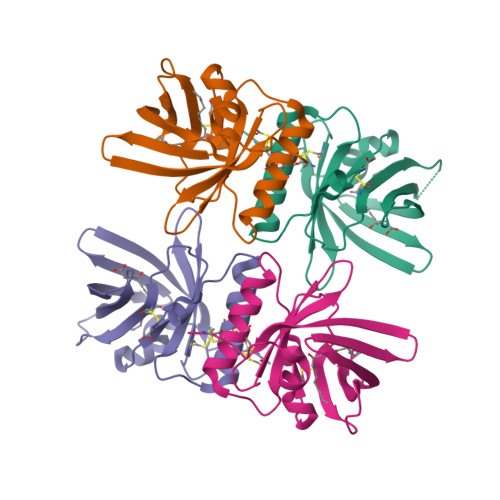
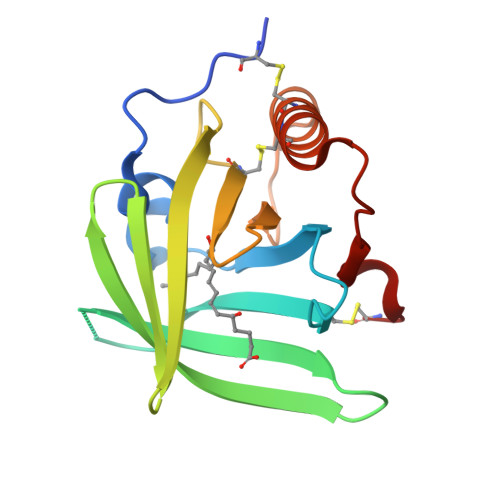
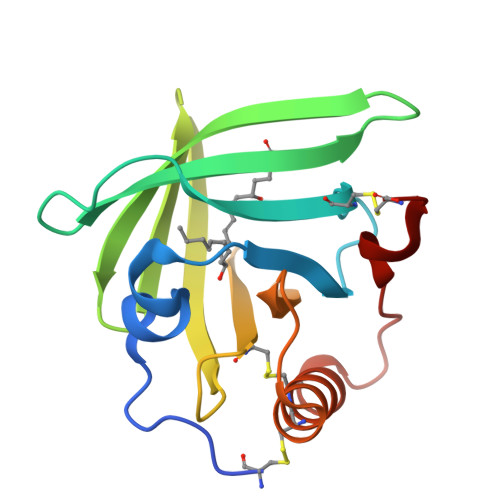
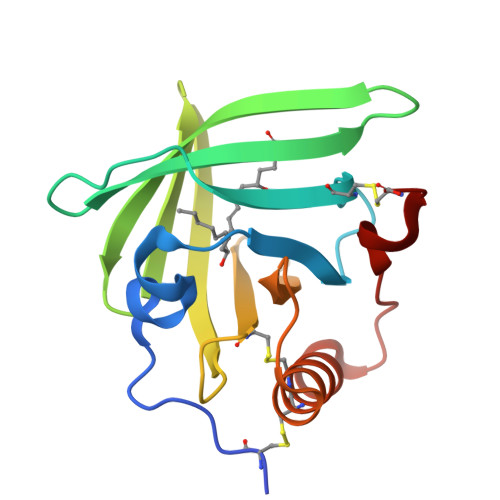
Bifunctional Lipocalin Ameliorates Murine Immune Complex-Induced Acute Lung Injury.
Roversi, P., Ryffel, B., Togbe, D., Maillet, I., Teixeira, M., Ahmat, N., Paesen, G.C., Lissina, O., Boland, W., Ploss, K., Caesar, J.J., Leonhartsberger, S., Lea, S.M., Nunn, M.A.(2013) J Biological Chem 288: 18789
- PubMed: 23625922
- DOI: https://doi.org/10.1074/jbc.M112.420331
- Primary Citation of Related Structures:
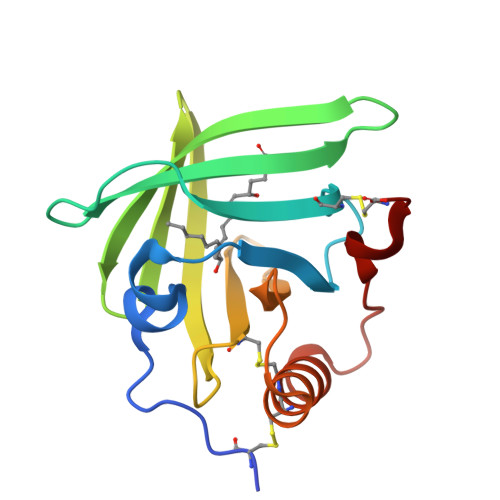
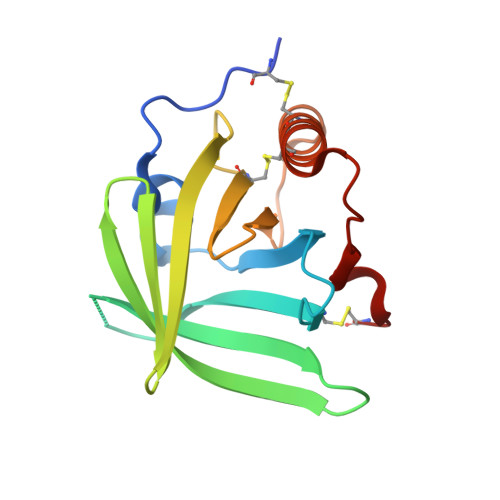
3ZUI, 3ZUO - PubMed Abstract:
Molecules that simultaneously inhibit independent or co-dependent proinflammatory pathways may have advantages over conventional monotherapeutics. OmCI is a bifunctional protein derived from blood-feeding ticks that specifically prevents complement (C)-mediated C5 activation and also sequesters leukotriene B4 (LTB4) within an internal binding pocket. Here, we examined the effect of LTB4 binding on OmCI structure and function and investigated the relative importance of C-mediated C5 activation and LTB4 in a mouse model of immune complex-induced acute lung injury (IC-ALI). We describe two crystal structures of bacterially expressed OmCI: one binding a C16 fatty acid and the other binding LTB4 (C20). We show that the C5 and LTB4 binding activities of the molecule are independent of each other and that OmCI is a potent inhibitor of experimental IC-ALI, equally dependent on both C5 inhibition and LTB4 binding for full activity. The data highlight the importance of LTB4 in IC-ALI and activation of C5 by the complement pathway C5 convertase rather than by non-C proteases. The findings suggest that dual inhibition of C5 and LTB4 may be useful for treatment of human immune complex-dependent diseases.
Organizational Affiliation:
Sir William Dunn School of Pathology, University of Oxford, Oxford OX1 3RE, United Kingdom.