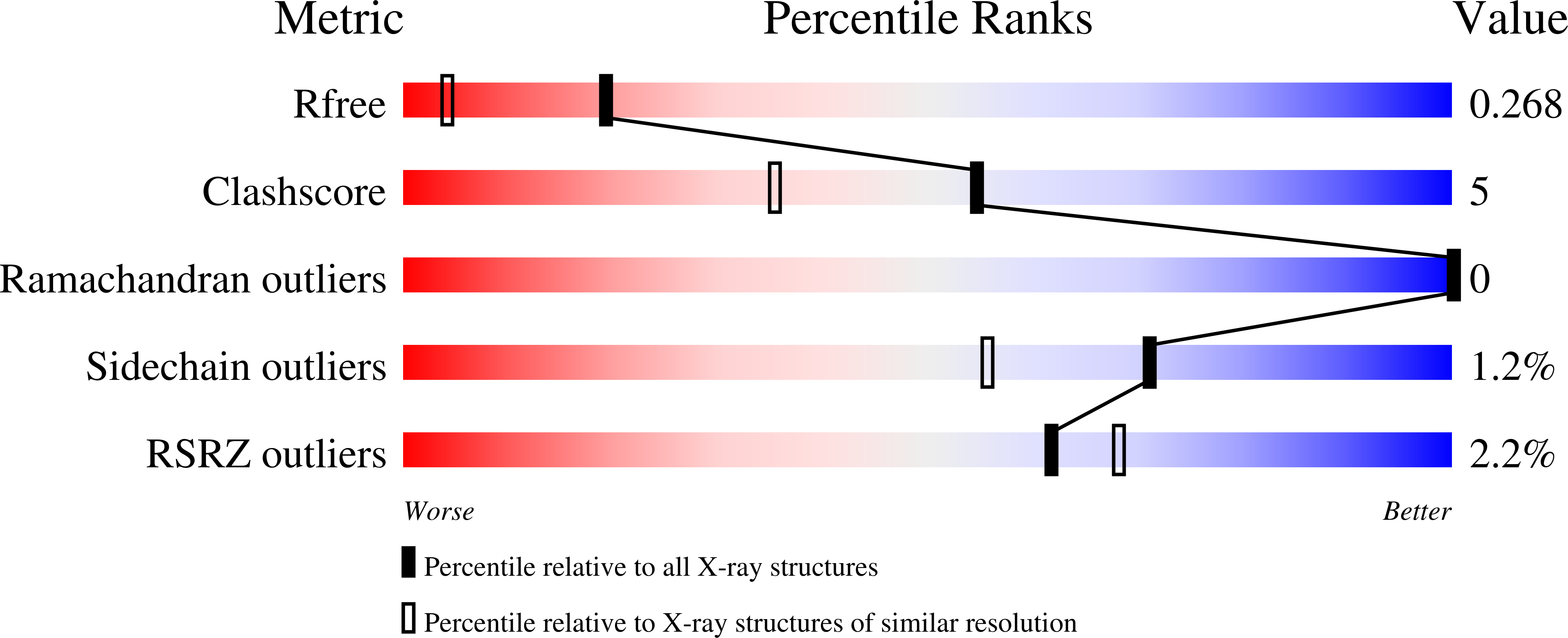
Covalent Small Molecule Inhibitors of Ca(2+)-Bound S100B.
Cavalier, M.C., Pierce, A.D., Wilder, P.T., Alasady, M.J., Hartman, K.G., Neau, D.B., Foley, T.L., Jadhav, A., Maloney, D.J., Simeonov, A., Toth, E.A., Weber, D.J.(2014) Biochemistry 53: 6628-6640
- PubMed: 25268459
- DOI: https://doi.org/10.1021/bi5005552
- Primary Citation of Related Structures:
4PDZ, 4PE0, 4PE1, 4PE4, 4PE7 - PubMed Abstract:
Elevated levels of the tumor marker S100B are observed in malignant melanoma, and this EF-hand-containing protein was shown to directly bind wild-type (wt) p53 in a Ca(2+)-dependent manner, dissociate the p53 tetramer, and inhibit its tumor suppression functions. Likewise, inhibiting S100B with small interfering RNA (siRNA(S100B)) is sufficient to restore wild-type p53 levels and its downstream gene products and induce the arrest of cell growth and UV-dependent apoptosis in malignant melanoma. Therefore, it is a goal to develop S100B inhibitors (SBiXs) that inhibit the S100B-p53 complex and restore active p53 in this deadly cancer. Using a structure-activity relationship by nuclear magnetic resonance approach (SAR by NMR), three persistent binding pockets are found on S100B, termed sites 1-3. While inhibitors that simultaneously bind sites 2 and 3 are in place, no molecules that simultaneously bind all three persistent sites are available. For this purpose, Cys84 was used in this study as a potential means to bridge sites 1 and 2 because it is located in a small crevice between these two deeper pockets on the protein. Using a fluorescence polarization competition assay, several Cys84-modified S100B complexes were identified and examined further. For five such SBiX-S100B complexes, crystallographic structures confirmed their covalent binding to Cys84 near site 2 and thus present straightforward chemical biology strategies for bridging sites 1 and 3. Importantly, one such compound, SC1982, showed an S100B-dependent death response in assays with WM115 malignant melanoma cells, so it will be particularly useful for the design of SBiX molecules with improved affinity and specificity.
Organizational Affiliation:
Center for Biomolecular Therapeutics (CBT), Department of Biochemistry and Molecular Biology, University of Maryland School of Medicine , Baltimore, Maryland 21201, United States.