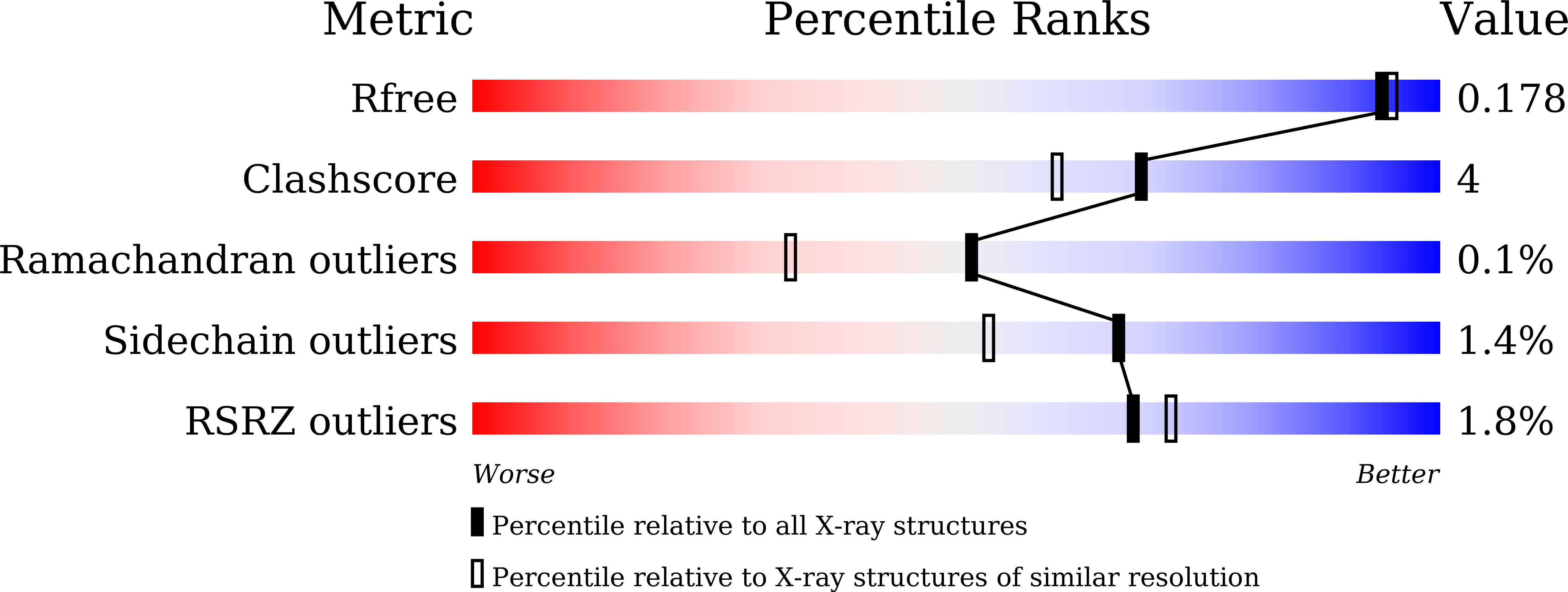
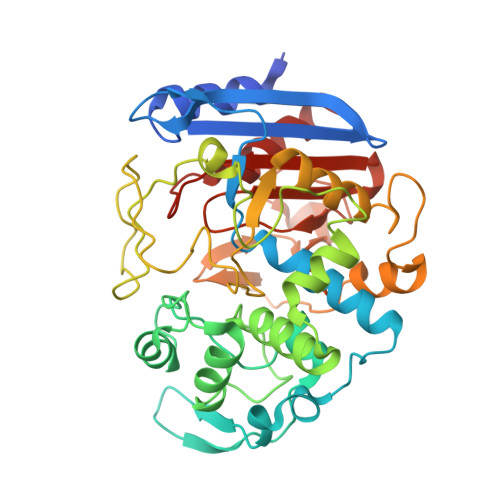
Structural Basis for the Enantioselectivity of Esterase Est-Y29 toward (S)-Ketoprofen
Ngo, T.D., Oh, C., Mizar, P., Baek, M., Park, K., Nguyen, L., Byeon, H., Yoon, S., Ryu, B.H., Kim, T.D., Yang, J.W., Seok, C., Lee, S.S., Kim, K.K.(2019) ACS Catal 9: 755-767