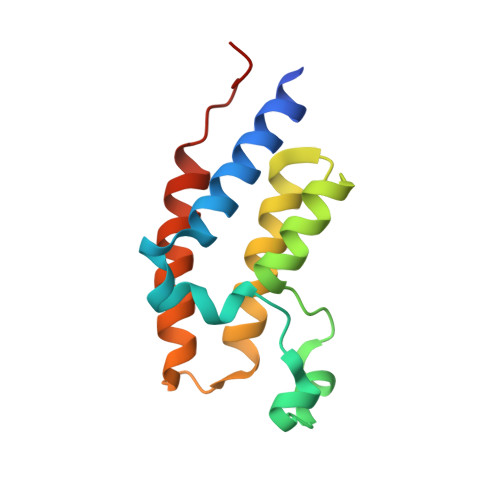
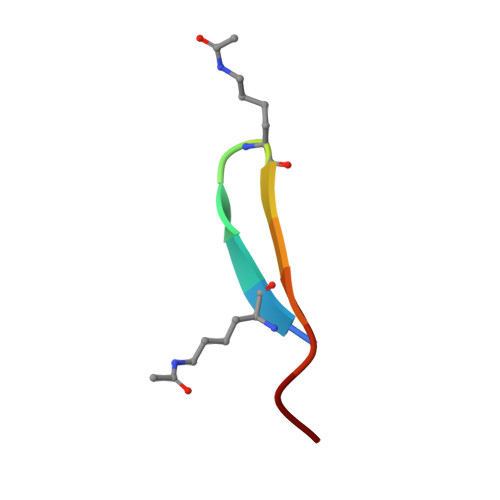
Discovery and characterization of cyclic peptides selective for the C-terminal bromodomains of BET family proteins.
Franck, C., Patel, K., Walport, L.J., Christie, M., Norman, A., Passioura, T., Suga, H., Payne, R.J., Mackay, J.P.(2023) Structure 31: 912-923.e4
- PubMed: 37269828
- DOI: https://doi.org/10.1016/j.str.2023.05.009
- Primary Citation of Related Structures:
8CV4, 8CV5, 8CV6, 8CV7, 8DNQ - PubMed Abstract:
DNA-encoded cyclic peptide libraries can yield high-potency, high-specificity ligands against target proteins. We used such a library to seek ligands that could distinguish between paralogous bromodomains from the closely related bromodomain and extra-terminal domain family of epigenetic regulators. Several peptides isolated from a screen against the C-terminal bromodomain of BRD2, together with new peptides discovered in previous screens against the corresponding domain from BRD3 and BRD4, bound their targets with nanomolar and sub-nanomolar affinities. X-ray crystal structures of several of these bromodomain-peptide complexes reveal diverse structures and binding modes, which nevertheless display several conserved features. Some peptides demonstrate significant paralog-level specificity, although the physicochemical explanations for this specificity are often not clear. Our data demonstrate the power of cyclic peptides to discriminate between very similar proteins with high potency and hint that differences in conformational dynamics might modulate the affinity of these domains for particular ligands.
- School of Chemistry, The University of Sydney, Sydney, NSW 2006, Australia; Australian Research Council Centre of Excellence for Innovations in Peptide and Protein Science, The University of Sydney, Sydney, NSW 2006, Australia.
Organizational Affiliation: